Empowering People to Manage Diabetes with a Unified Mobile App
Living with diabetes means daily tracking and constant care. SEP helped a global medical device company simplify that experience with a unified mobile app that turns blood glucose data into clear, actionable insights for users worldwide.
Opportunity
People everywhere use our client’s mobile tools to track diabetes. Whether on iOS or Android, users need a reliable, easy-to-use app, and our client needed to deliver updates faster.
Services
Results
- 5× user growth worldwide
- 30+ languages, 120+ countries
- Faster FDA-compliant releases
Client
A Global Healthcare Leader Advancing Diabetes Care
Our client is one of the world’s largest pharmaceutical companies and a long-standing innovator in healthcare. Founded more than 125 years ago, the organization combines strengths in pharmaceuticals and diagnostics to deliver transformative solutions across major disease areas.
Within its broad portfolio, the company provides glucose monitoring systems, insulin delivery devices, and digital tools that empower people with diabetes—along with their caregivers and healthcare providers—to better manage daily care, wherever they are on their journey.
About the Client
Industry
Healthcare
Type
Public
Market
Global
Employees
10,000+

Opportunity
Managing Two Apps Slowed Progress for Millions of Users
The client’s diabetes management app connected to Bluetooth® glucose meters and served users around the world. But maintaining separate Android and iOS codebases made updates complex and time-consuming—a critical issue for software regulated by the FDA.
They needed a faster, more reliable way to deliver new features and improvements without compromising safety or user experience.
What We Did
CI/CD
Cross-Platform Development
Maintenance
Mobile Development
Product Localization
Usage Analytics
Solution
From Two Codebases to One Reliable Platform
SEP analyzed the existing mobile apps, user workflows, and regulatory requirements, then recommended moving to a unified cross-platform approach. This gave the client a single, shared codebase that supported both iOS and Android while still allowing platform-specific features where needed.
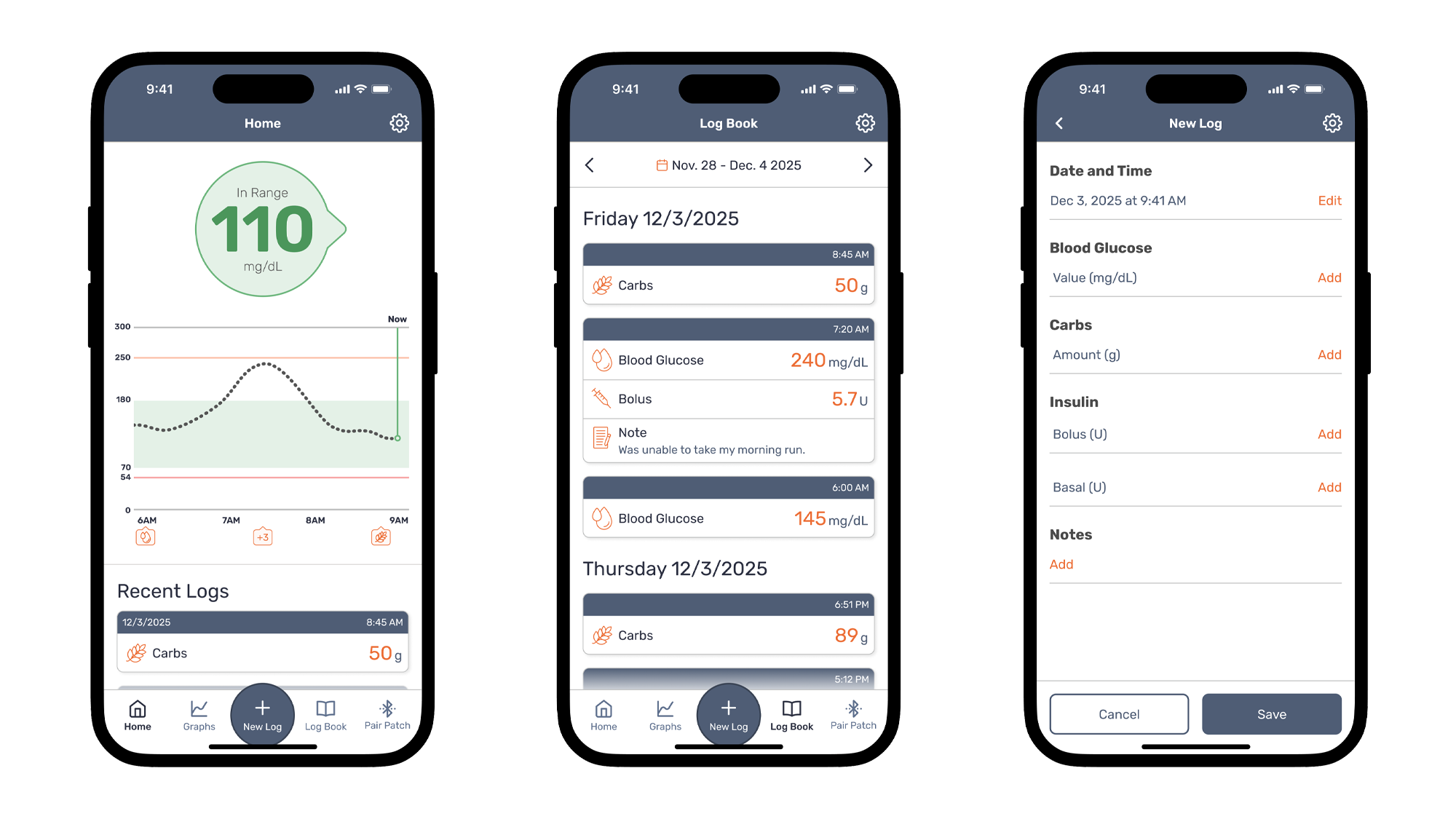
To begin the transition, SEP used the client’s existing Android Java code as a foundation and converted it into C#, bringing both apps into one consolidated environment. The team rebuilt each feature, validated Bluetooth® connectivity to glucose meters, and ensured a consistent experience for users on both platforms.
This unified approach simplified testing, reduced long-term maintenance costs, and created a stronger foundation for analytics, compliance, and future updates.
Tech Stack
Results
Analytics That Improve Care
With one shared C# codebase for Android and iOS, SEP helped the client cut duplication, reduce risk, and release updates faster—vital for FDA-regulated software. Usage analytics improved dramatically, showing how users interact with the app and helping teams resolve field issues more quickly. Since launch, global adoption has increased fivefold, and more than one million glucose readings are synced every month.
5× increase in global app users
1M+ glucose readings logged monthly
30+ languages across 120+ countries
Faster, safer software releases
Reduced testing duplication
Enhanced analytics for field insights